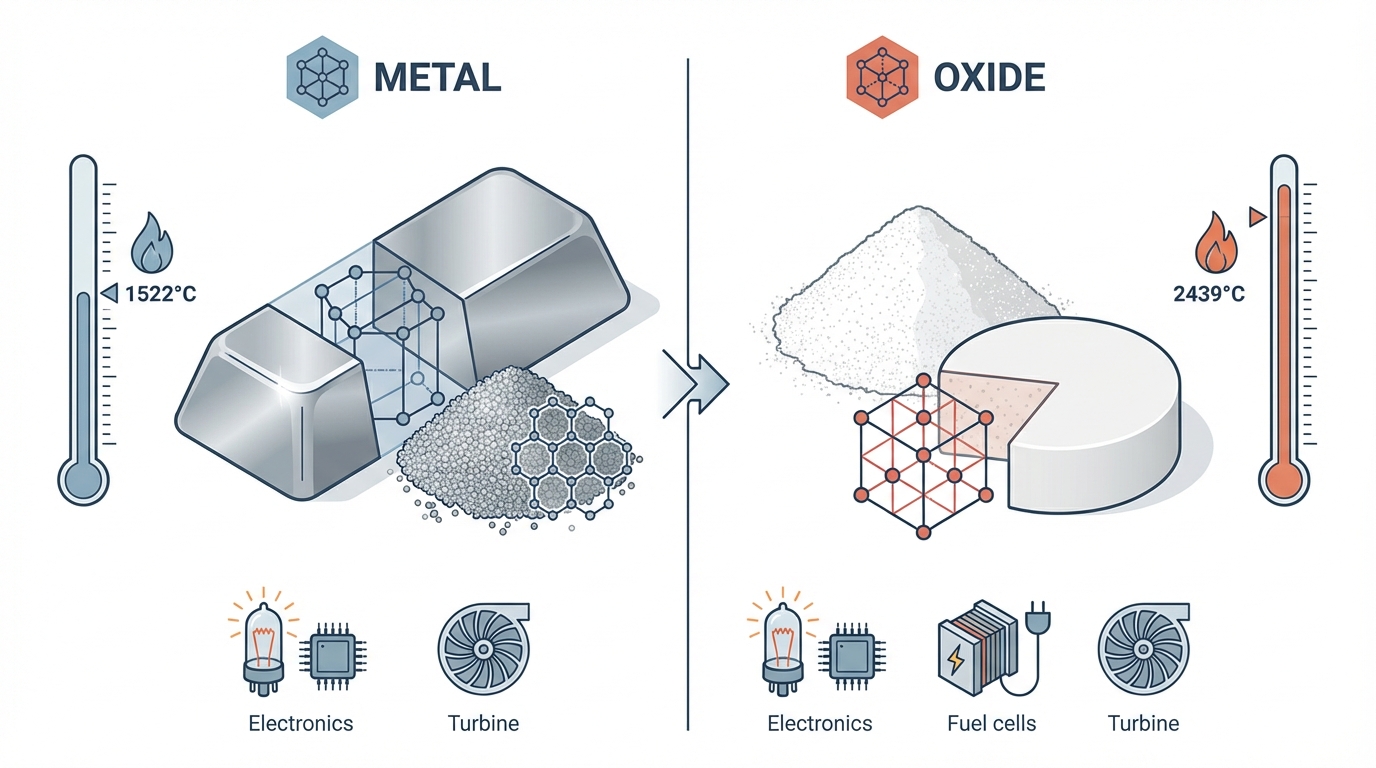
Yttrium Metal vs Oxide in 2024‑2025: Fast Technical Reference for Sourcing and Operations
Executive context. Yttrium (Y, atomic number 39) occupies an unusual position in the rare earths system: the oxide (Y₂O₃) underpins high‑volume applications in electronics, energy ceramics, and optics, while the metal serves much smaller but highly sensitive roles in aerospace and defense alloys. Physically and logistically, these are almost two different commodities. The oxide is a stable white powder moving in bulk; the metal is a reactive, sometimes pyrophoric solid that demands inert handling and specialised freight. That divergence shapes everything from process design to export‑control exposure.
Publicly available technical notes and industry commentary indicate that most yttrium demand in 2024‑2025 flows through oxide into phosphors, ceramic electrolytes, and laser hosts, while metal tonnage remains comparatively small but mission‑critical for turbine components, specialty welding, and some defense‑grade superalloys.[1-7] The industrial question is no longer simply “is yttrium available,” but rather: which form, at what purity, with which processing and compliance path, and under what geopolitical and ESG constraints.
The sections below anchor that assessment on four pillars: the metal-oxide property split, the process routes that transform ore into usable forms, the application clusters that lock in demand, and the specific bottlenecks that now dominate supply chain risk.
1. Fundamental Properties: Why Metal and Oxide Behave Like Different Commodities
1.1 Yttrium metal: reactive, ductile, and logistics‑sensitive
Yttrium metal is a silvery transition metal with a hexagonal close‑packed crystal structure at room temperature and a density of about 4.47 g/cm³.[2,4] Its melting point near 1526°C and boiling point around 3336°C support use in high‑temperature environments, particularly as a microalloying element in nickel, magnesium, and aluminium systems where creep resistance and oxidation behaviour matter.
In air, yttrium rapidly forms a thin protective oxide film. That passivation is beneficial for component lifetimes but comes with a critical caveat: fine metal powders and turnings can become pyrophoric. Industry datasheets report a risk of spontaneous ignition of metal fines above roughly 400°C in air, which has pushed standard practice toward storage in inert atmospheres (argon) and vacuum‑sealed packaging for higher‑surface‑area forms.[2,6] For bulk logistics, that translates into hazardous‑material classification, segregated storage, and in many cases a bias toward specialist carriers rather than generic bulk freight.
From a processing perspective, the metal’s main handicap is its reactivity. Producing high‑purity (>99.9% or 99.99% by mass) yttrium metal typically requires high‑temperature reduction and molten‑salt electrolysis, both of which carry non‑trivial energy intensity and limited batch sizes, particularly when purity specifications include sub‑10‑ppm thresholds for transition metal contaminants in advanced alloys or superconducting applications.
1.2 Yttrium oxide (Y₂O₃): ceramic workhorse for electronics and energy
In contrast, yttrium oxide (Y₂O₃) is a chemically stable, white ceramic powder with a cubic crystal structure, a melting point around 2425°C, and a relatively high refractive index in the 1.9-2.1 range.[1,5] It is robust against moisture and oxygen, does not ignite, and can be shipped as a standard industrial chemical in bulk bags or drums. Its optical and dielectric properties, as well as its ability to form solid solutions with zirconia and other oxides, underpin its dominant role in:
- phosphors for LEDs and displays (typically Y₂O₃ doped with rare earth activators such as europium),
- yttria‑stabilized zirconia (YSZ) electrolytes and coatings,
- laser host materials and transparent ceramics, and
- high‑performance refractories and crucibles.
Oxide powders can be processed by conventional ceramic routes-spray drying, pressing, and sintering around 1000–1200°C-or incorporated as dopants into alumina, zirconia, and garnet structures. Because of this process familiarity, most industrial demand is specified directly in oxide form, with purity grades such as 99.5% (for general ceramics and phosphors) or 99.9% (for optics and electronics) that are accessible at industrial scale.
1.3 Property comparison and immediate supply implications
| Property | Yttrium Metal | Yttrium Oxide (Y₂O₃) | Supply Chain Implication |
|---|---|---|---|
| Physical form | Ingot, rod, granules, powder (reactive) | Stable white powder | Oxide suitable for bulk shipping; metal often handled as hazmat |
| Density | ≈ 4.47 g/cm³ | ≈ 5.0 g/cm³ | Oxide denser; influences volumetric loading in phosphors and ceramics |
| Melting point | ≈ 1526°C | ≈ 2425°C | Oxide suited to ultra‑high‑temperature ceramics; metal to high‑T alloys |
| Reactivity | Passivates, but powders can be pyrophoric | Non‑flammable, chemically robust | Metal requires inert storage; oxide fits conventional warehouse regimes |
| Typical purity specs | >99.9–99.99% for advanced alloys, superconductors | ≈99.5%+ for phosphors and ceramics | Metal production effectively batch‑limited; oxide can be produced continuously |
| Relative cost level (2024 industry commentary) | Several times higher per kg than oxide | Baseline cost reference | Metal pricing dominated by processing and compliance overhead |
Industry briefs frequently characterise yttrium as “oxide‑first”: for most high‑volume applications, material flows as Y₂O₃, not as metal.[1,3,5] Metal production is reserved for cases where yttrium must be incorporated into metallic matrices (superalloys, aluminium/magnesium alloys, or specialty solders), or where direct electrochemical reduction is integral to a process. This bifurcation is the first structural element of yttrium’s risk profile.
2. Process Routes: From Ore to Oxide to Metal
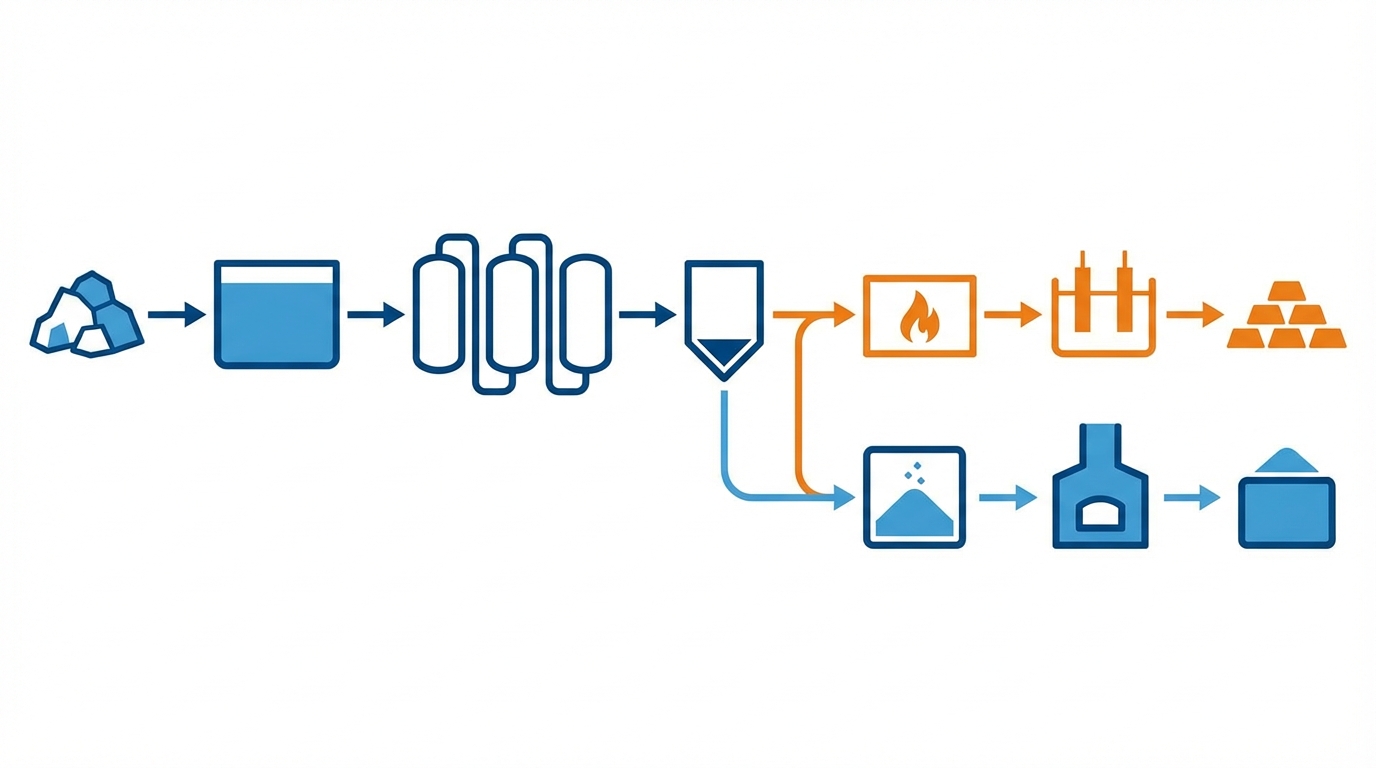
2.1 Upstream: ores, clays, and mixed rare earth streams
Yttrium rarely occurs in isolation. It is typically recovered as part of a mixed rare earth element (REE) stream from:
- Bastnäsite and monazite ores, where yttrium is associated with light and some heavy rare earths, often at grades on the order of fractions of a percent to a few percent TREO (Total Rare Earth Oxides).
- Ionic adsorption clays in southern China and parts of Southeast Asia, where yttrium and heavy rare earths are loosely bound to clay surfaces and can be leached with weak acid solutions.[2,7]
In both cases, the mining and beneficiation steps-crushing, grinding, and flotation—are optimised to generate a mixed REO concentrate rather than a yttrium‑rich stream per se. Typical flowsheets target a rare‑earth oxide concentrate with elevated heavy‑REE content, which only later becomes a yttrium fraction through solvent extraction and precipitation. That shared upstream means yttrium volumes are tightly coupled to broader REE mining decisions and to demand for co‑products such as neodymium, dysprosium, and terbium.
2.2 Midstream: chemical separation and oxide refining
Once in solution—typically after sulphuric or hydrochloric acid leaching—the mixed rare earth stream enters large‑scale solvent extraction (SX) circuits. For yttrium, extractants such as phosphoric or phosphonic acids (e.g., P507) in organic diluents are arranged in long counter‑current banks, often comprising hundreds of mixer‑settler stages, to gradually separate light and heavy REEs and then individual elements.[2,6]
Yttrium frequently tracks with heavier rare earths; the point in the SX train where a reasonably pure yttrium stream is available depends on feed composition and process tuning. Downstream, yttrium is precipitated (often as an oxalate), filtered, and calcined to Y₂O₃ at 1000–1200°C. This route yields oxide purities in the 99–99.9% range with recoveries that industry literature often describes as “in the 90%+ band” for well‑run circuits, but at the cost of significant reagent consumption and energy use.[2,8]
The key industrial reality: SX banks are capital‑intensive, complex to operate, and highly tuned to specific feed blends. Retuning a circuit to change the relative output of yttrium versus other REEs is not instantaneous; it means extended campaigns, intermediate recycles, and often modifications to stripping and precipitation sections. That inertia underpins the lag between price signals and changed oxide availability.
2.3 Downstream: oxide‑to‑metal conversion and its constraints
Transforming Y₂O₃ into metallic yttrium usually follows a two‑stage route:
- Calciothermic reduction: Y₂O₃ is mixed with calcium and sometimes other additives, then heated at elevated temperatures. The simplified reaction Y₂O₃ + 3Ca → 2Y + 3CaO captures the basic chemistry.[2] Industrial practice must manage side reactions, control oxygen activity, and handle the resulting CaO‑rich slag. Reported metal purities from this stage generally fall below the final specifications for aerospace or superconducting uses, necessitating further refining.
- Molten‑salt electrolysis: Yttrium chloride salts (often YCl₃ in a LiCl or similar melt) are electrolysed at temperatures around 800°C in inert atmospheres.[6] Electrolytic cells are typically operated in batches—commonly cited in the tens of kilograms scale—constrained by anode/cathode degradation, contamination control, and energy management.
Each step introduces yield losses and waste streams (for example, calcium‑rich slag and spent salts) that attract ESG scrutiny and disposal costs. Industry commentary frequently notes that oxide‑to‑metal conversion has significantly higher specific energy consumption than oxide production itself and that effective recoveries after trimming to tight purity specs are noticeably lower than headline chemical yields.
This combination—batch electrolysis, tight impurity limits, and hazardous‑material handling—explains why the global yttrium metal market remains measured in hundreds of tonnes per year, while oxide moves in the tens of thousands of tonnes. The bottleneck is technological and regulatory, not geologic.
3. Application Clusters and How They Lock in Form Requirements
Yttrium demand in the literature is often segmented into four broad clusters: electronics and displays, energy systems, aerospace and defense, and optics/medical uses.[1,3,5,7] Each cluster is anchored in either oxide or metal form, with different purity and microstructural constraints, which in turn determine exposure to specific parts of the supply chain.
3.1 Electronics and displays: phosphors and dielectric ceramics
In displays and solid‑state lighting, yttrium almost exclusively enters as oxide. The classic example is Y₂O₃:Eu red phosphors, where Y₂O₃ is doped with europium to convert UV or blue excitation into red emission for LED packages and flat‑panel displays.[1,5] These formulations demand tight control of particle size, dopant distribution, and impurity levels (notably transition metals that can quench luminescence).
Because performance is highly sensitive to trace contaminants, phosphor producers commonly specify oxide in the 99.5–99.9% purity range with narrow limits on iron, nickel, and other coloured ions. Once production lines are tuned around a specific powder morphology and purity profile, switching suppliers or feed origins involves non‑trivial requalification and potentially full re‑binning of LED performance grades. That process rigidity is one reason display‑grade Y₂O₃ frequently carries a premium over more generic ceramic grades.
Beyond phosphors, yttrium‑containing dielectric ceramics appear in capacitors and substrates where a balance between permittivity, breakdown strength, and thermal stability is required. Again, these are oxide‑centric uses; metallic yttrium plays no direct role at the component level.
3.2 Energy systems: YBCO superconductors and YSZ fuel cells
Energy applications highlight yttrium’s role in advanced functional materials:
- High‑temperature superconductors such as YBa₂Cu₃O₇₋ₓ (YBCO) rely on yttrium at the A‑site of the perovskite‑like lattice. Here, Y₂O₃ is consumed as a precursor in powder synthesis and thin‑film deposition. Critical current performance at liquid‑nitrogen temperatures (~77 K) is sensitive to stoichiometry and impurity levels, prompting demands for high purity and controlled oxygen content.[2,5]
- Yttria‑stabilised zirconia (YSZ) for solid oxide fuel cells (SOFCs) and gas turbine components typically contains around 8–10 mol% Y₂O₃ in zirconia. This stabilises the cubic or tetragonal phase and enables high oxide‑ion conductivity at operating temperatures often cited in the several‑hundred‑to‑around‑800°C range.[5,6]
For SOFCs, the electrolyte and interconnect materials are among the highest value‑add uses of yttrium oxide by mass. Public energy roadmaps in Europe and Asia describe aggressive deployment targets for fuel cells in power and industrial applications, implying sustained demand for Y₂O₃‑bearing ceramic powders and coatings. YSZ production is oxide‑intensive; conversion to metal is irrelevant here and would simply add cost and energy consumption.
Electrodes and interconnects in some fuel cell and hydrogen technologies may integrate Ni‑Y or other yttrium‑enriched alloys, where the metal form re‑enters the picture. In those cases, traceability of metal feed and compliance with dual‑use regulations (in particular when alloys overlap with defense specifications) become part of the operational picture.
3.3 Aerospace and defense: small tonnages, high strategic sensitivity
Aerospace and defense applications concentrate much of the strategic attention on yttrium, despite relatively modest tonnage. Two families dominate:
- Microalloyed metals. Small additions of yttrium (often on the order of 0.1–1% by weight) to nickel‑based superalloys, aluminium, or magnesium can improve high‑temperature creep resistance, grain boundary cohesion, and oxide scale adherence.[3,4] These improvements are particularly relevant for turbine blades, combustor components, and structural alloys in high‑heat zones.
- Thermal barrier and environmental coatings. Yttrium oxide is a key dopant in some zirconia‑based thermal barrier coatings (TBCs) applied by plasma spraying or electron‑beam physical vapor deposition (EB‑PVD). These coatings slow thermal degradation of turbine parts and can extend service intervals materially when engineered correctly.[3]
Defense procurement documents cited in industry analyses describe yttrium metal purity specs at or above 99.9% for certain jet engine and airframe components, with additional restrictions on trace elements that affect fatigue and oxidation behaviour. This narrows the supplier pool to a small set of qualified refiners capable of delivering consistent analysis (often verified by techniques such as glow‑discharge mass spectrometry, GDMS). Export controls (e.g., ITAR/EAR in the United States) can apply not only to finished alloys but also, in some cases, to specific high‑purity input streams.
The result is a two‑tier system: commodity‑like oxide supply for generic ceramics and phosphors, and a much more tightly controlled metal and high‑purity oxide channel for aerospace and defense. Disruptions to the latter may not show up easily in aggregate REE statistics but can halt specific engine or system programs.
3.4 Optics, lasers, and medical use
Optical and medical uses add a further layer of specificity to yttrium demand:
- Laser hosts. Yttrium aluminium garnet (YAG, Y₃Al₅O₁₂) and related structures form the backbone of many industrial lasers. Here, Y₂O₃ is consumed into high‑purity alumina‑yttria mixtures that are melted and grown into single crystals, often by the Czochralski process, over multi‑day cycles.[2,4] Defect rates on the order of fractions of a percent can already impact yields for high‑power laser rods, putting a premium on powder cleanliness and process control.
- Transparent ceramics. Fully dense polycrystalline Y₂O₃ or YAG can serve as infrared windows and laser hosts when porosity and grain size are tightly managed. This segment is extremely sensitive to powder morphology, sintering aids, and contamination.
- Medical isotopes. Radioisotopes such as yttrium‑90 are used in radiotherapy; while the source chemistry may involve oxides or salts, the volumes are small compared with industrial ceramics but demand very high radiochemical purity and controlled decay logistics.[4,7]
In these domains, yttrium is typically locked into long‑term qualification chains: once a laser platform or radiopharmaceutical process is validated with a specific yttrium chemistry and supplier, substitution requires fresh regulatory and technical validation. That significantly reduces short‑term elasticity of demand.
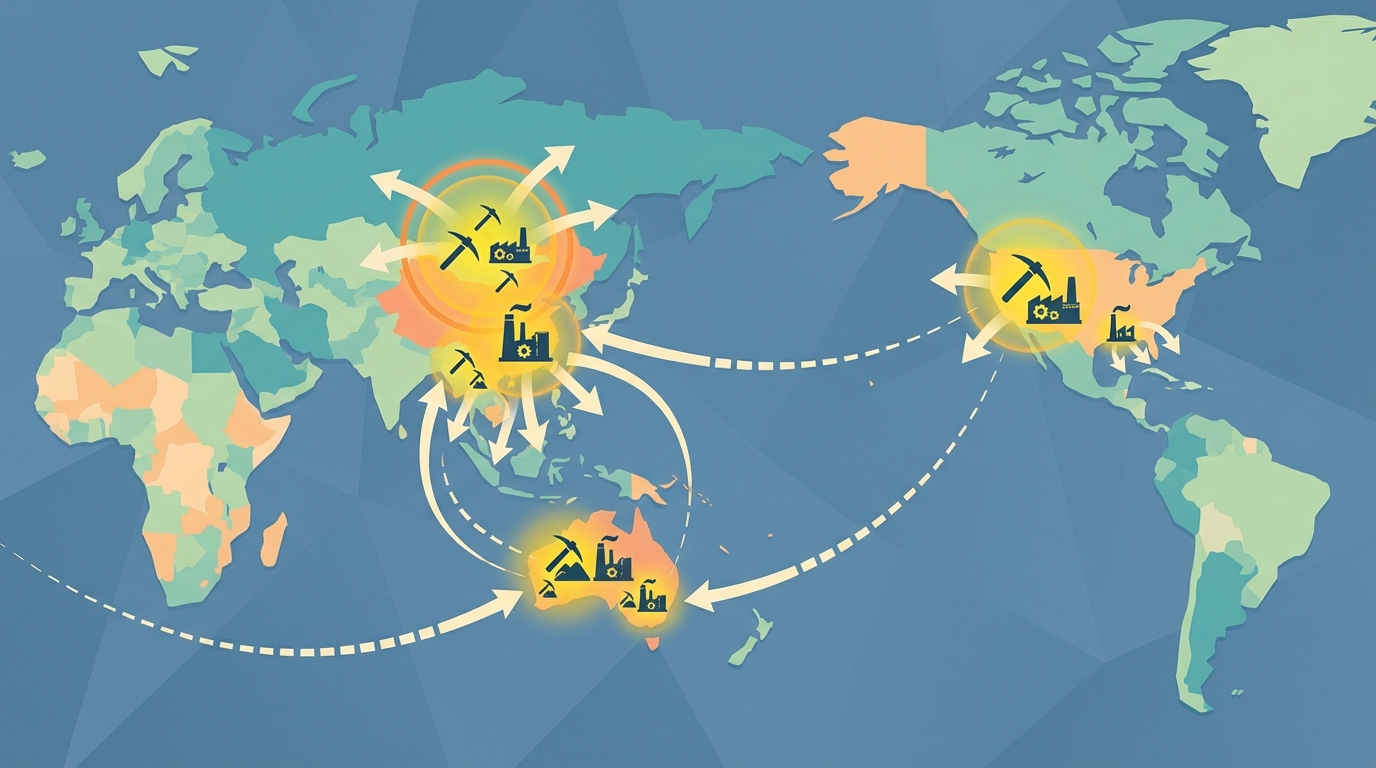
4. Global Supply Architecture and Geographic Concentration
Public sources including the USGS, company disclosures, and specialist commodity analyses consistently highlight that China dominates yttrium oxide refining capacity and a large share of mining, particularly through the Bayan Obo complex in Inner Mongolia and ionic clay operations in the south.[3,7,8] A limited number of non‑Chinese actors—most prominently Lynas (Australia/Malaysia) and MP Materials (United States)—have begun to bring meaningful non‑Chinese yttrium oxide volumes to market, though typically as part of broader REE product slates rather than as stand‑alone yttrium plays.
Metal production is even more concentrated, with a small set of Chinese refineries historically handling most oxide‑to‑metal conversion and a handful of pilot or early‑stage projects in North America and elsewhere exploring domestic metallisation routes, often co‑funded under defense or critical‑minerals initiatives.[3,8]
| Form | Representative Producers (2024–2025) | Indicative Capacity (MT/year) | Status | Primary Constraint Highlighted in Public Sources |
|---|---|---|---|---|
| Yttrium metal | Ganzhou‑area refiners (China) | Several hundred | Operational | Export licensing and allocation; purity control for aerospace grades |
| Yttrium metal | U.S. pilot plants (e.g., defense‑linked projects) | Tens (pilot scale) | Development / early operations | Scale‑up, molten‑salt cell durability, and permitting |
| Y₂O₃ oxide | Chinese REE processors (e.g., Baotou, Longnan regions) | Thousands | Operational | Export quotas, environmental compliance, tailings management |
| Y₂O₃ oxide | Lynas (Australia / Malaysia) | Several hundred (design scale) | Operational / ramping | Feed composition, heavy‑REE balance, and downstream qualification |
| Y₂O₃ oxide | MP Materials (U.S.) | Building from tens toward hundreds | Ramping | Integration of separation capacity and customer qualification |
Several structural features stand out:
- Concentration of refining. While geological occurrences of yttrium are more widespread, practical refining capacity for high‑purity Y₂O₃ remains heavily concentrated in China. Non‑Chinese plants are still establishing long operating histories and qualification track records.
- Metal as a niche within a niche. Metal production is not just a subset of oxide production; it depends on dedicated high‑temperature and electrochemical infrastructure that only a few sites operate at scale.
- Co‑product dynamics. Because yttrium is usually co‑produced with other REEs, increases in yttrium output require either higher total REE throughput or a re‑optimised separation train. Both are capital‑ and time‑intensive.
In practical terms, this means that sourcing strategies anchored in non‑Chinese oxide and domestically metallised feed remain in a building phase through 2025. For downstream sectors—especially those with strict defense or critical‑infrastructure classifications—that lag is a core operational consideration.
5. Process Bottlenecks and Operational Risk Points
5.1 Separation and refining bottlenecks
The most technically demanding step in yttrium’s journey is often not mining but separation. Multi‑hundred‑stage solvent extraction circuits blur the boundaries between chemistry and continuous‑flow engineering. Common failure modes observed in industry case studies include:
- Organic phase degradation under oxidative or thermal stress, leading to loss of extractant performance and contamination.
- Phase disengagement issues as feed composition drifts, which can cause carry‑over of impurities and reduce stage efficiency.
- Throughput‑purity trade‑offs, where pushing higher flow reduces residence time and degrades separation, particularly for closely neighbouring heavy REEs.
Yttrium’s position among heavy rare earths means that small drifts in SX performance can have disproportionate effects on Y₂O₃ purity or recovery, especially when circuits are also tuned for dysprosium or terbium extraction. Plants targeting high‑purity Y₂O₃ often accept lower throughput or more recycles to maintain target specs, raising operating costs and lengthening lead times.
5.2 Oxide–metal conversion and purity specifications
On the metal side, calciothermic reduction and molten‑salt electrolysis introduce their own bottlenecks:
- Current efficiency and cell life. Elevated temperatures (~800°C) and reactive halide melts place mechanical and chemical stress on electrodes and linings. Electrolyser downtime for maintenance or relining directly caps annual output.
- Impurity control. Calcium, oxygen, chlorine, and transition metals all need to be controlled within tight bands for aerospace and superconducting applications. Every refining step that tightens impurity windows sacrifices some yield.
- Scale limitations. Industry descriptions of batch sizes in the 50–100 kg class underscore how far yttrium remains from bulk metals such as aluminium or copper in process scale. Scaling out via multiple cells is possible but multiplies capital and maintenance requirements.
One practical implication: “oxide‑first” sourcing followed by just‑in‑time metallisation looks attractive on paper but can collide with real‑world electrolysis capacity limits and qualification bottlenecks for high‑purity metal.
5.3 Environment, safety, and regulatory pressure points
Environmental and safety regulation increasingly shapes yttrium’s operational landscape:
- Tailings and waste management. Rare earth separation plants generate tailings containing low‑level radioisotopes and chemical residues. Several publicised incidents at REE plants in China and elsewhere have tightened local enforcement and increased effective downtime for inspections and remediation.[3,8]
- Impurity limits in products. Frameworks such as EU REACH are progressively lowering allowable heavy‑metal impurities in chemicals, including yttrium oxide. Meeting these limits can require more aggressive purification, higher reagent use, and more sophisticated waste treatment.
- Hazard classification. Yttrium metal powders and turnings can fall under flammable solid or spontaneously combustible classifications, constraining allowable packaging, warehouse conditions, and freight modes. That classification propagates through insurance and transport‑planning decisions.
These factors do not eliminate yttrium flows, but they introduce additional sources of unplanned downtime, capex for mitigation, and permitting risk—particularly for new non‑Chinese projects seeking social licence to operate.
5.4 Logistics friction and route vulnerability
Because yttrium volumes are moderate but highly value‑dense, logistics choices tend toward containerised sea freight for oxide and a mix of sea and air for higher‑value oxide and metal. Several friction points stand out:
- Metal freight constraints. Hazardous‑material labelling for pyrophoric‑risk forms reduces the pool of carriers and aircraft that will accept yttrium metal, especially in powdered or granular form. This can lengthen booking lead times and increase freight rates compared with oxide.
- Route disruptions. Deviations around constrained maritime corridors can add weeks to shipping times between Asian producers and European or North American consumers. For just‑in‑time operations, that effectively widens safety‑stock requirements.
- Port and customs scrutiny. Dual‑use concerns and export‑control regimes drive more detailed paperwork and occasional inspections for high‑purity metal and certain oxide grades, particularly when end‑uses intersect with defense or advanced electronics.
One revealing observation from logistics data is that yttrium metal does not behave like a standard minor metal from a freight perspective; it behaves more like a specialty chemical or energetically reactive material, with all the scheduling and compliance overhead that implies.
6. Observed Sourcing Patterns and Trade‑offs in 2024–2025
6.1 Oxide‑centric strategies in electronics and optics
In LEDs, displays, and optical ceramics, most downstream actors continue to treat high‑purity Y₂O₃ as the primary control lever rather than considering metal. Several trends are visible in public disclosures and industry interviews:
- Diversification of oxide origin. There is visible interest in qualifying yttrium oxide from non‑Chinese refiners such as Lynas and emerging Western projects, even at a unit‑cost premium, to reduce single‑country exposure for critical phosphor and ceramic feedstocks.[3,5]
- Tighter long‑term supply agreements. Where material functions are hard to substitute (e.g., specific phosphor blends or laser‑host powders), longer‑term contracts and collaborative process optimisation are becoming more common, trading some price flexibility for continuity and joint quality control.
- Segmentation by grade. Commodity‑grade oxide for generic ceramics is increasingly decoupled from higher‑spec electronics/optics grades, with distinct supplier sets and qualification paths.
The operative trade‑off in this segment tends to be cost versus geopolitical and quality risk, rather than oxide versus metal form. The metal route does not compete technically with high‑purity oxide for these applications.
6.2 Defense and aerospace: preference for traceable metal and controlled oxide streams
Defense‑linked aerospace programs place particular emphasis on domestic or allied‑origin yttrium streams, both for metal and for certain high‑purity oxides entering thermal barrier coatings and advanced ceramics. Public documents associated with national defense authorizations and critical‑minerals strategies highlight:
- funding for pilot and demonstration plants capable of oxide‑to‑metal conversion within national borders,
- qualification of specific refiners against stringent impurity specifications for turbine and structural alloys, and
- closer integration between mining, separation, and metallisation to improve traceability and reduce dependence on foreign intermediary refiners.[3,7,8]
These efforts reflect a strategic calculus: although total metal tonnage is modest, availability of certified, traceable, high‑purity yttrium metal and oxide can become a single‑point failure for particular propulsion or sensor systems. In this niche, the premium for secure metal supply is a function of industrial resilience rather than commodity pricing.
6.3 Energy technologies: managing YSZ and superconductor supply
Growth in SOFC deployments, hydrogen infrastructure, and advanced power systems has put additional attention on YSZ and YBCO precursor supply. Observed patterns include:
- Integrated YSZ supply chains. Several YSZ producers source yttrium oxide via long‑term arrangements with REE refiners, sometimes with back‑to‑back clauses tying volumes to publicly announced fuel‑cell or turbine projects.
- Specification‑driven sourcing. SOFC electrolyte formulations often have narrow windows for Y₂O₃ content (e.g., 8–10 mol%) and impurity levels. Changes in oxide origin that affect minor impurities, particle morphology, or surface chemistry can require requalification of entire stack designs.
- Regional balancing. In regions pursuing aggressive decarbonisation policies, there is growing interest in localising at least part of the YSZ and superconductor feedstock chain to reduce dependency on geopolitically exposed supply routes.
Here again, oxide remains the dominant form; metal demand is limited to specific alloying roles in ancillary components. The main trade‑offs revolve around security of high‑spec oxide supply versus cost and flexibility.
6.4 Risk‑management practices observed across the chain
Across sectors, several operational risk‑management patterns around yttrium have become visible in 2024–2025, framed explicitly as industrial resilience rather than financial optimisation:
- Enhanced material qualification and testing. High‑spec users commonly rely on techniques such as ICP‑MS for oxide and GDMS for metal to verify that delivered product meets declared purity and impurity profiles, particularly for new suppliers or origins.
- Upstream traceability and ESG audits. There is growing attention to mine‑of‑origin data, REE balance, tailings practices, and community impacts, both for compliance with emerging regulations and for reputational risk management.
- Scenario analysis around export controls and quotas. Some downstream actors model multi‑month disruptions to specific producing countries or refiners, assessing the impact on production schedules and identifying points where alternative suppliers or temporary design changes might be technically viable.
- Form diversification. In limited cases, holding a portion of yttrium as oxide and a portion as pre‑qualified metal has been used to buffer against conversion bottlenecks or sudden shifts in regulatory treatment of particular forms.
These practices reflect a recognition that yttrium’s most material risks arise where technical specifications, process bottlenecks, and geopolitical controls intersect, rather than from headline tonnage alone.
7. Conclusion: Yttrium as a Lever Point in the Rare Earths System
Yttrium illustrates how a single element can sit at the crossroads of ceramics, alloys, and advanced functional materials while remaining structurally constrained by midstream processing and regulatory overlays. The oxide/metal split effectively creates two parallel markets: a larger, more commodity‑like oxide market dominated by Chinese refining, and a smaller, tightly specified metal and high‑purity oxide segment that underpins aerospace, defense, and critical energy systems.
Technically, the hardest problems are no longer in mining but in solvent extraction, oxide purification, and molten‑salt electrolysis. These unit operations define recoveries, purities, and throughput, and they are where most ESG, safety, and permitting pressures apply. “More ore” does little to alleviate short‑term risk unless it connects to separation and metallisation capacity that meets end‑use specifications.
For engineering, procurement, and compliance teams, the structure of yttrium’s processing chain explains why disruptions at a single SX complex or metallisation plant can ripple into apparently distant sectors such as MRI magnets, turbine blades, or industrial lasers. Once specific Y₂O₃ powders or Y‑containing alloys are embedded in qualified designs, substitution ceases to be a simple sourcing decision and becomes a multi‑year technical program.
From TI22 Strategies’ perspective, yttrium sits among the more subtle but consequential levers in the broader rare earths system: small in tonnage but large in strategic impact. Ongoing analysis therefore tracks not only headline production data but also weak signals in separation technology, molten‑salt process scale‑up, ESG regulation, and export‑control policy that are likely to redefine yttrium’s risk profile over the coming years.
Note on the TI22 methodology TI22 Strategies combines systematic monitoring of regulatory texts (including export control updates and environmental mandates), high‑frequency trade and production data, and close reading of technical specifications in downstream sectors such as turbines, fuel cells, and semiconductor equipment. This cross‑view allows early identification of mismatches between qualifying material requirements and the evolving capabilities and constraints of yttrium mining, separation, and metallisation infrastructure.
Sources and further reading
- Honrel – “Top 10 Applications of Yttrium Oxide”[1]
- Stanford Advanced Materials – “Yttrium: Element Properties and Uses” and related technical notes[2,6]
- Tradium – Yttrium industry product data and application briefs[3]
- AEM REE – Yttrium facts and application overviews[4]
- Parchem – “Yttrium Oxide Uses in Electronics, Ceramics and Energy Applications”[5]
- EBSCO / research‑starter entries on yttrium geology and uses[7]
- AZoM – “Yttrium – Properties and Applications” and related property databases[8]
- USGS – “Rare Earths Statistics and Information” (yttrium within REE statistics)
- Lynas and MP Materials – public operations updates and technical disclosures on REE separation, including yttrium‑bearing streams